Some reactions that do not qualify as acid-base reactions under the other definitions do so under only the Lewis definition. 
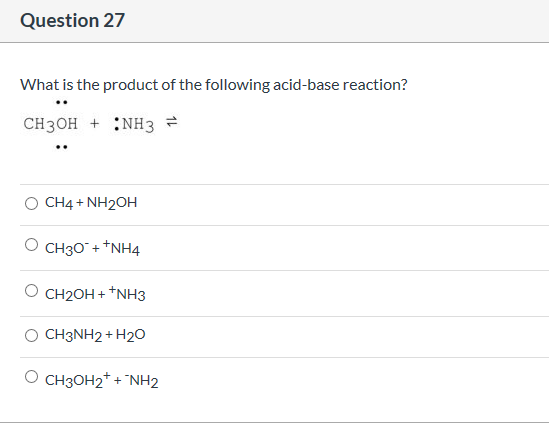
(CC BY-NC CK-12) The lone pair on the nitrogen atom is transferred to the hydrogen ion, making the NH 3 a Lewis base while the H + is a Lewis acid. The dissociation of an acid HA can be represented as: HA > A- + H+. 1: Reaction between ammonia and a proton. Acid-base reaction is the transfer of a proton from an acid to a base. A base is a species which accepts or tends to accept a proton. 
The reaction is usually written as: (1) N H X 3 + H C l N H X 4 C l. An acid is defined as a species (a compound or an ion) which donates or tends to donate a proton (II+ ion). The ammonium ion is the conjugate acid of the base ammonia, NH 3 its acid ionization (or acid hydrolysis) reaction is represented by NH 4 + ( a q) + H 2 O ( l) H 3 O + ( a q) + NH 3 ( a q) K a K w / K b Since ammonia is a weak base, Kb is measurable and Ka > 0 (ammonium ion is a weak acid). In chemistry, neutralization or neutralisation (see spelling differences), is a chemical reaction in which an acid and a base react quantitatively with each other. The reaction between ammonia and hydrogen chloride to give ammonium chloride is a neutralization reaction, since ammonia acts as a base and hydrogen chloride acts as an acid. Salts are composed of related numbers of cations (positively charged ions) and anions (negative ions) so that the product is electrically neutral (without a net charge). In chemistry, a salt is an ionic compound that can be formed by the neutralization reaction of an acid and a base. In chemistry, bases are substances that, in aqueous solution, are slippery to the touch, taste astringent, change the color of indicators (e.g., turn red litmus paper blue), react with acids to form salts, promote certain chemical reactions (base catalysis), accept protons from any proton donor, and/or contain completely or partially displaceable OH− ions. Question : Is NH3 an acid or base or neutral ?Īn acid is a molecule or ion capable of donating a hydron (proton or hydrogen ion H+), or, alternatively, capable of forming a covalent bond with an electron pair (a Lewis acid).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
